The CARES Act specifies that sunscreens that conform to these requirements (and to the other requirements specified by section 505G of the FD&C Act, including the general requirements for nonprescription drugs) are deemed to be GRASE and not new drugs. Section 505G of the FD&C Act, which was enacted by the CARES Act, created a “final administrative order” for sunscreens (the deemed final order) consisting of the requirements specified in the 1999 stayed (not in effect) final monograph for OTC sunscreen products, except for requirements governing labeling and effectiveness, which are described in a final labeling and effectiveness testing rule for sunscreens published in 2011. Specifically, the CARES Act amended the Federal Food, Drug, and Cosmetic (FD&C Act) to replace the monograph rulemaking process with an administrative order process for issuing, revising, and amending OTC monographs. On March 27, 2020, the Coronavirus Aid, Relief, and Economic Security Act (CARES Act) was signed into law. The CARES Act includes statutory provisions to reform and modernize the way over-the-counter (OTC) monograph drugs are regulated in the United States. Why did the FDA issue a deemed final order for over-the-counter (OTC) sunscreen products and then issue a proposed order shortly after?Ī. 
The following questions and answers provide more information on the deemed final order and proposed order. The proposed order reflects FDA’s proposed requirements for OTC sunscreen products for the future. FDA also posted the proposed order for sunscreens to amend and revise this deemed final order for OTC sunscreens products. FDA posted the deemed final order for sunscreens which sets the current requirements for marketing OTC sunscreen products. And while one might think that a recall is a no-brainer, things are a bit muddled because the FDA “does not define a specific limit for drugs like sunscreen.On September 24, 2021, FDA took steps aimed at improving the quality, safety, and efficacy of sunscreens as part of its implementation of new authorities for certain over-the-counter (OTC) drugs. 
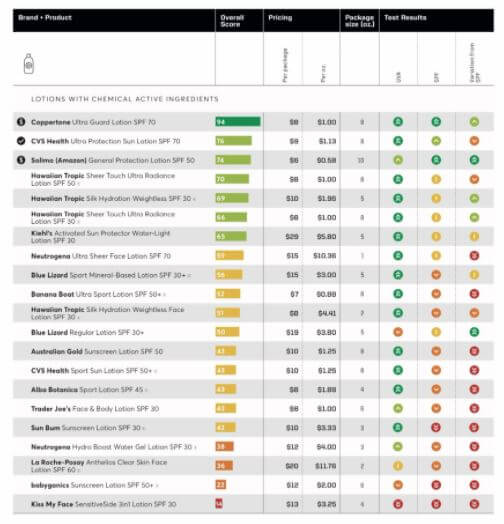
Valisure is urging the FDA to recall the above products and created a petition to support its efforts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
